Of almost 4000 retractions listed in the Retraction Watch database in 2021, over a quarter involved “concerns/issues about image” or “duplication of image.” Similarly, a recent study found that two-thirds of 120,000+ comments on PubPeer involve concerns about images. Inappropriate manipulations range from innocent errors to “beautification” to intentional falsification. It is unsurprising, then, that many journals now use specialised image screeners to look for signs of inappropriate manipulation. In this blog we discuss current guidelines on processing biomedical images and question how much processing is too much.
Digital images should be minimally processed.
This guideline is the cornerstone of image processing – image data is (arrays of numerical) data and should be treated as such. Altering numerical data is considered unacceptable, yet many think it is permissible to clean up image data. Generally, “the intention is not to deceive” but, rather, to simplify the data to make it more understandable to the reader.
Cleaning up backgrounds

In this example, we (the blog writers) have erased data using image editing software. Figure taken from FigShare (FigShare dataset ID: 12495611, version 1). While erasing data like this may not affect the overall interpretation, it is considered bad practice. The erased data may not be relevant to the study in question. However, it may be “real and biologically important” to some readers, or its interpretation may change over time as more information becomes available.
Cropping
It is common practice to remove irrelevant parts of a gel or western blot; however, it is unacceptable to crop an image in such a way that misrepresents the data or cuts out essential information. Many journals encourage authors to submit unprocessed whole gels for review, or at least that they include a certain amount of area above and below the band. “A reasonable guide is to retain about five bandwidths of background above and below.” Readers may be suspicious of tight cropping as it suggests the authors may be trying to hide another band nearby. Images that will be compared should be cropped to the same size.
Splicing
Splicing images together should be avoided where possible. If it is not possible to run an experiment on a single gel, for example, if you have numerous samples, it may be necessary to splice together data from two different gels. Splicing should be clearly indicated in the image (usually with a white space or black line between splices) and described in the figure legend. Bands should not be spliced together to look like they are from the same gel.
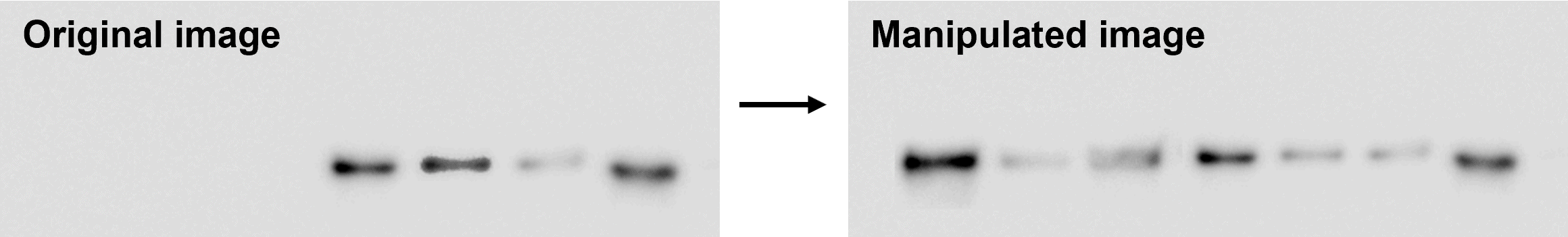
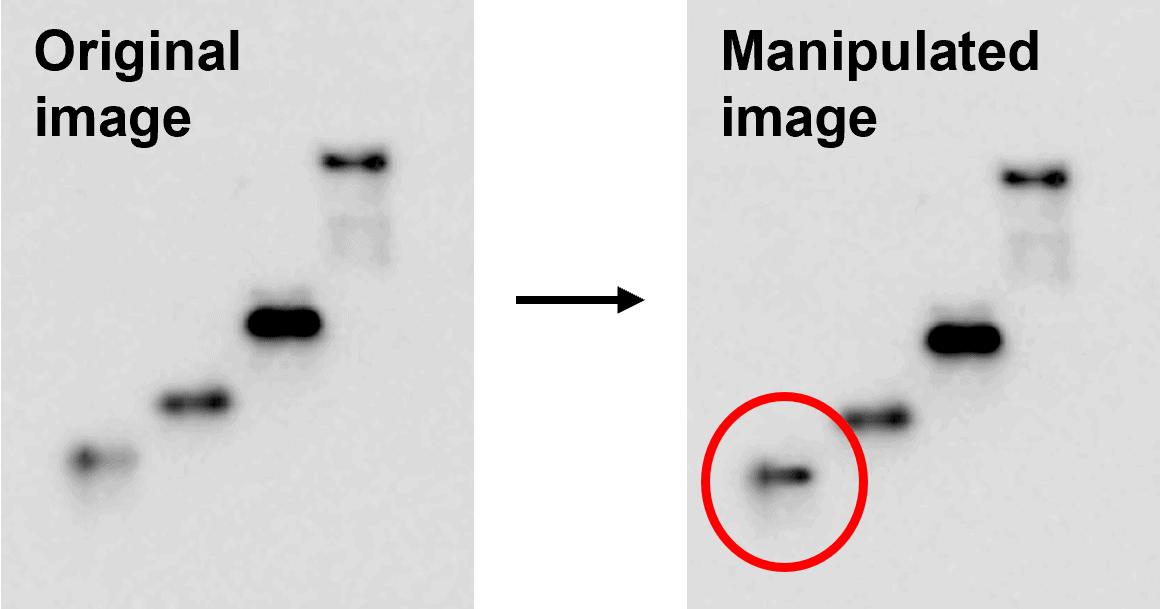
In this example, we have juxtaposed bands from different gels together to give the impression that they are from the one gel. Figure taken from FigShare (FigShare dataset ID: 12495611, version 1).
Simple adjustments
Simple adjustments—for example, brightness/contrast, which are applied to every pixel in an image—may be applied uniformly to an entire image; however, not to the extent that data are obscured or lost entirely.
Adjustments to enhance or obscure specific features within an image should be avoided. To emphasize a specific feature, use arrows or pseudocoloring where appropriate.
In this example, we have modified a band to give the impression of more even intensity. Figure taken from FigShare (FigShare dataset ID: 12495611, version 1).
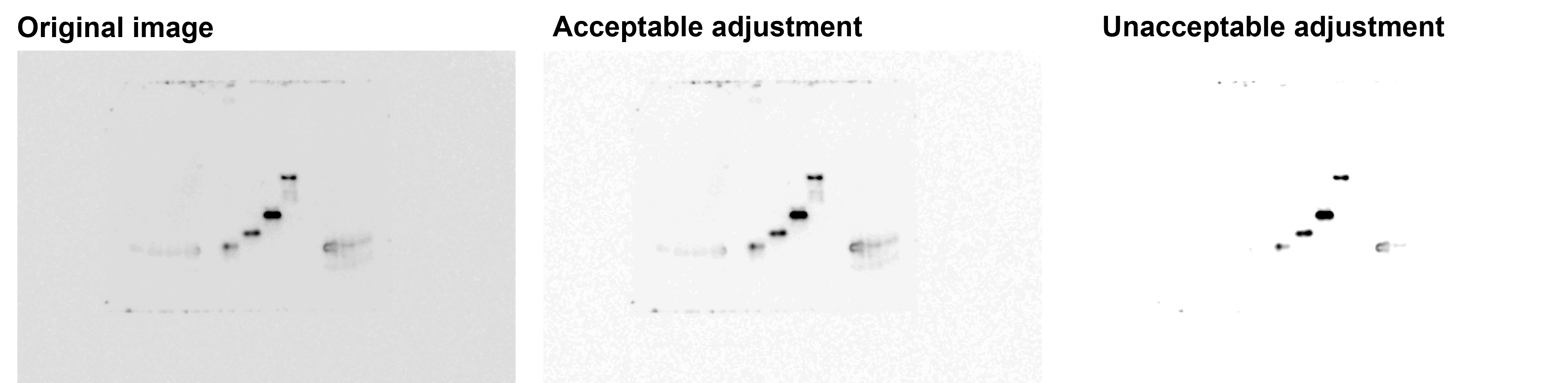
Adjustments should be applied uniformly to images that will be compared to one another.
In this example, adjustments to the exposure have not been applied uniformly for all images. Figure taken from FigShare (Figure 4, DOI: 10.1371/journal.pone.0042720). This is a misrepresentation of what the author originally observed.
Loading controls
Loading controls should be run through the gel from the same loading well as the experimental sample you want to probe. If this is not possible, it should be disclosed and justified in the figure legend. It is unacceptable to duplicate loading control blots for different experiments. “The point of a loading control is to be able to see how consistently samples were loaded onto and run/transferred through the same gel. It is against this control that changes in levels of the band of interest are calibrated.”
Creative micrographs
You may be tempted to combine interesting features from different microscope fields into one micrograph, especially if there are restrictions on the number of figures your target journal allows. However, this practice is unacceptable.
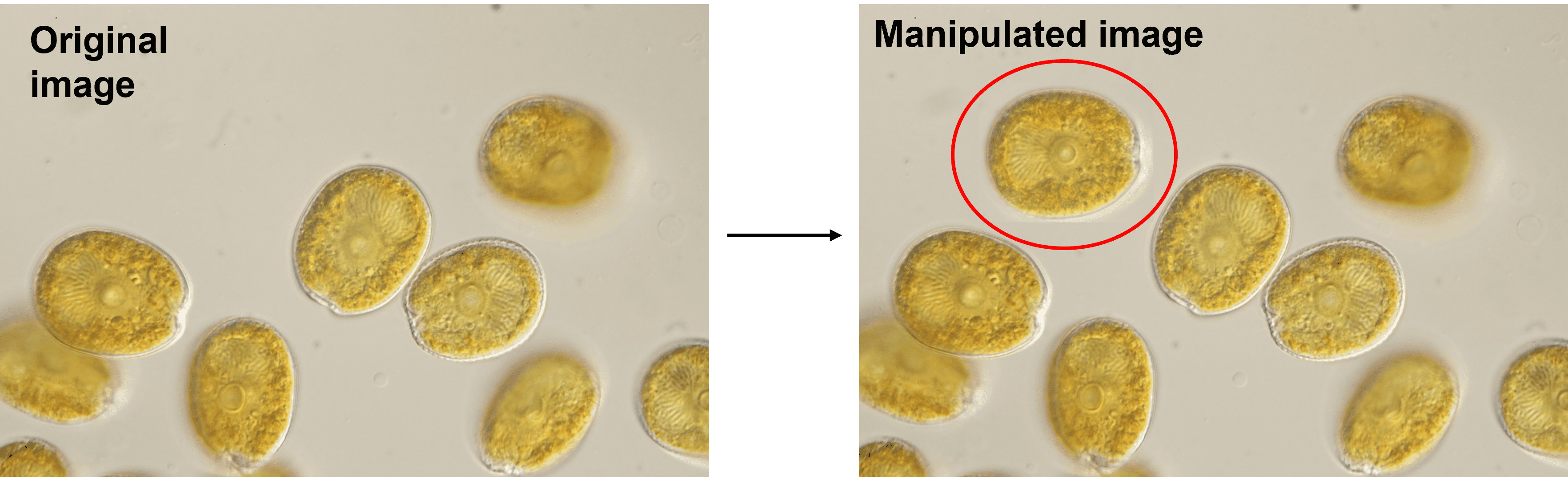
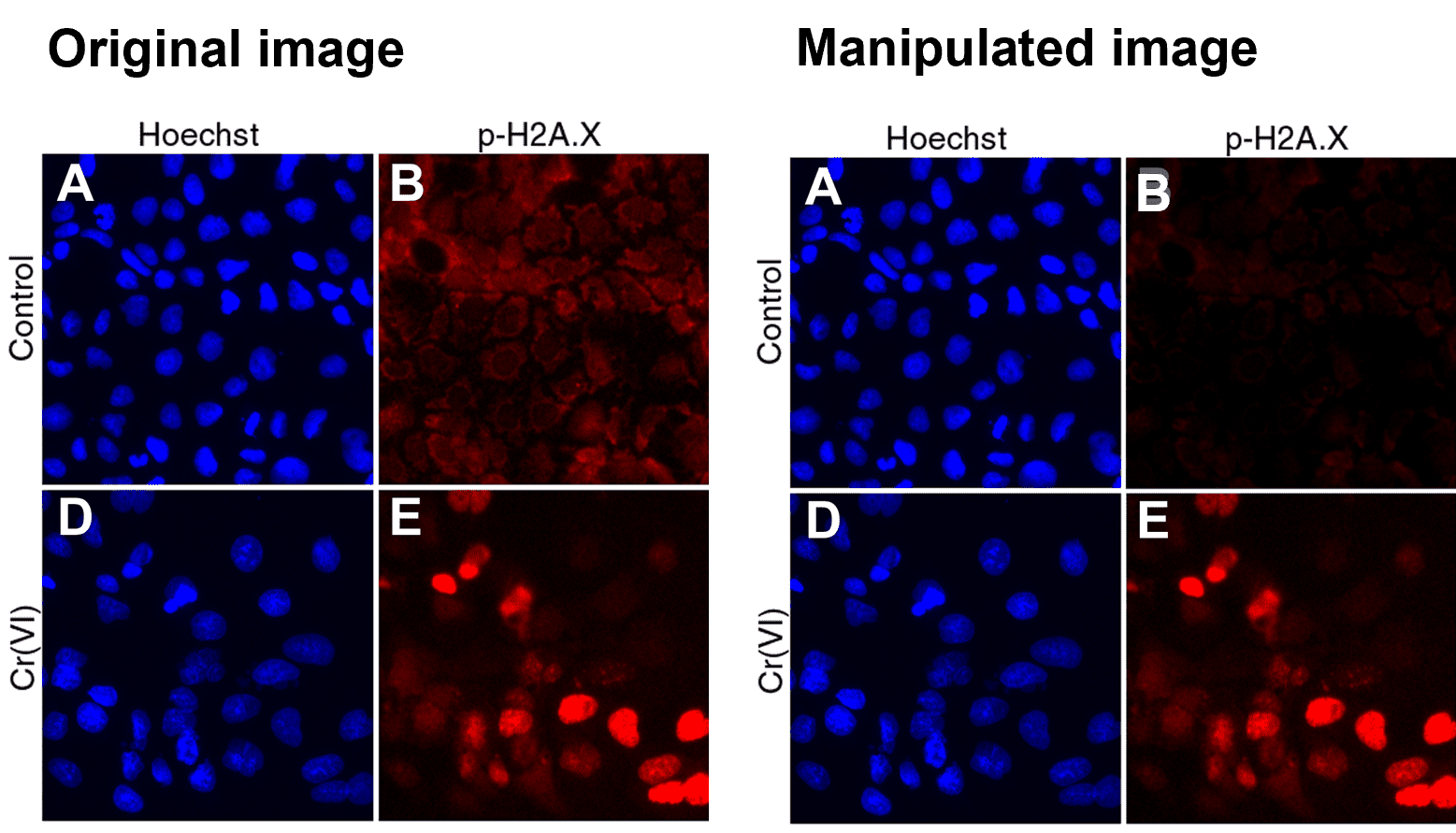
In this example, we have combined features from two separate microscope fields into one. Figure taken from FigShare (FigShare dataset ID: 1254461, version 2).
If you make a mistake
It can be very tempting to fix a mistake using image processing, for example, if you loaded too much protein on a gel resulting in overly saturated bands. Digitally adjusting the bands to appear less saturated may have no effect whatsoever on the overall interpretation of the data; however, it is not an accurate representation of what you observed.
Every effort should be made to obtain the highest quality images possible at the data collection stage. Producing beautiful images for publication may require multiple repeat experiments and considerable scientific skill. The reader expects this level of care and effort.
Non-linear adjustments
Non-linear adjustments—for example, image gamma, which involves changes to only certain pixels in an image—should be described and justified in the figure legend or materials and methods. For example,
The gamma settings were adjusted to 0.45 on the green channel only. This was necessary to show the relevant structures of the glial cell bodies clearly.
Processing should only be done on a copy
Image processing should be performed on a copy of the unprocessed RAW image data files. You should always retain the unprocessed original image data. This could help you avoid a delayed publication or even an embarrassing correction or retraction.
Conclusion
From Rossner and Yamada’s seminal paper on this topic:
Data must be reported directly, not through a filter based on what you think they “should” illustrate to your audience. For every adjustment that you make to a digital image, it is important to ask yourself, “Is the image that results from this adjustment still an accurate representation of the original data?” If the answer to this question is “no,” your actions may be construed as misconduct.
Further reading
Cromey DW. Digital images are data: and should be treated as such. In Cell Imaging Techniques 2012 (pp. 1-27). Humana Press, Totowa, NJ. Available from: Digital Images Are Data: And Should Be Treated as Such (nih.gov)
Rossner M, Yamada KM. What’s in a picture? The temptation of image manipulation. Journal of Cell Biology. 2004 Jul 5;166(1):11-5. Available from: What’s in a picture? The temptation of image manipulation | Journal of Cell Biology | Rockefeller University Press (rupress.org)
